Magda El-Shenawee
https://news.uark.edu/articles/41165/researchers-move-closer-to-improved-method-of-detecting-breast-cancer
FAYETTEVILLE, Ark. – Engineering researchers at the University of Arkansas have moved closer to developing an alternative method of detecting and possibly treating breast cancer.
The researchers, led by Magda El-Shenawee, professor of electrical engineering, work with pulsed, terahertz imaging, a type of electromagnetic radiation technology previously used to find land mines. They adapted the technology to detect tumors and provide highly specific images of them.
Standard breast cancer imaging techniques do not always provide clear assessment of breast tissue on the margins of a tumor. Without an accurate picture of the margins between the tumor and healthy tissue, surgeons cannot be sure they have removed the entire tumor during a surgery.
This shortcoming contributes to high rates – 20 to 40 percent – of secondary surgery, either lumpectomy or mastectomy. Terahertz imaging could lead to fast, noninvasive, and highly specific tumor margin assessment, which in turn could reduce the occurrence of second surgeries, cancer reoccurrence and metastasis.
FINDINGS
In their most recent study, funded by a $424,081 grant from the National Institutes of Health, the researchers created terahertz images of breast adenocarcinoma cells, a type of malignancy, excised from mice. These images were taken from 13 tumor samples. To test the accuracy of the terahertz method, the images were then statistically compared to high-resolution histopathology images of the same excised tumor samples. Histopathology is the microscopic examination of tissue changes caused by disease.
In all tissue samples containing only cancer and fat – a combination similar to tissue in the human breast – the terahertz images, when compared to the histopathology images, accurately detected cancerous tissue with a high level of specificity.
On the other hand, samples containing cancer, fat and muscle showed only a reasonable correlation, El-Shenawee said. In these samples, the terahertz images detected cancer cells at the margins of tumors and muscle tissue, but not at high enough levels of specificity.
“Overlap between muscle and cancer tissue in the terahertz image creates some challenge in correctly classifying these regions,” El Shenawee said. “While muscle is unlikely to be present in surgical sections of human breast cancer, other kinds of fibrous tissue may be, so this requires further investigation with more advanced models.”
Future work will focus on spontaneously generated breast cancer tumors from genetically modified mice, which have tumor and tissue structures closer to that of humans. El-Shenawee said these models will provide a more accurate assessment of terahertz imaging. The researchers will also compare the terahertz images to other standard imaging techniques, such as radiography and computed tomography, or CT scan.
TERAHERTZ IMAGING
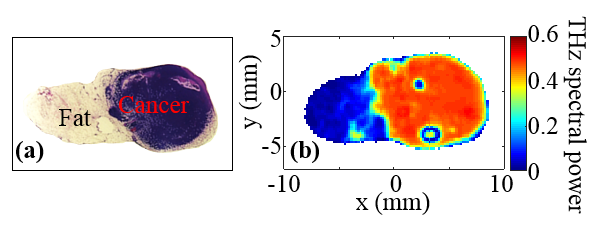
Pathology image, left, and corresponding terahertz image, right, of excised tissue from mouse breast tumor. Photo submitted by the researcher. |
Pulsed, terahertz spectroscopy produces high-quality images of the tissue, down to 80 micrometers. It scatters fewer waves than radiography, which enables deeper imaging into an object. Also, because terahertz radiation can transmit through most non-metallic materials, the systems can “see” through concealing barriers. For many years, El-Shenawee has focused on developing this detection system for health-care applications, while also investigating the unique electromagnetic signals emitted by breast cancer cells.
THE TEAM
The research team included Tyler Bowman, Tanny Chavez, graduate students in electrical engineering; Jingxian Wu, associate professor of electrical engineering; Kamrul Khan, graduate student in mathematical sciences; Avishek Chakraborty, assistant professor of statistics; Narasimhan Rajaram, assistant professor of biomedical engineering; and Keith Bailey, animal pathologist at Oklahoma State University.
About the College of Engineering: The University of Arkansas College of Engineering is the largest engineering program in the state of Arkansas. Over the past decade, the college has experienced unprecedented growth. Undergraduate enrollment has doubled since 2007, and total enrollment in the college is now over 4,000 students. The College of Engineering offers graduate and undergraduate degrees in nine engineering fields, as well as incorporating distance learning and interdisciplinary programs. Faculty in the college conduct research in many key areas, including electronics, energy, biomedical and healthcare engineering, materials science, transportation and logistics.
About the University of Arkansas: The University of Arkansas provides an internationally competitive education for undergraduate and graduate students in more than 200 academic programs. The university contributes new knowledge, economic development, basic and applied research, and creative activity while also providing service to academic and professional disciplines. The Carnegie Foundation classifies the University of Arkansas among only 2 percent of universities in America that have the highest level of research activity. U.S. News & World Report ranks the University of Arkansas among its top American public research universities. Founded in 1871, the University of Arkansas comprises 10 colleges and schools and maintains a low student-to-faculty ratio that promotes personal attention and close mentoring.









